|
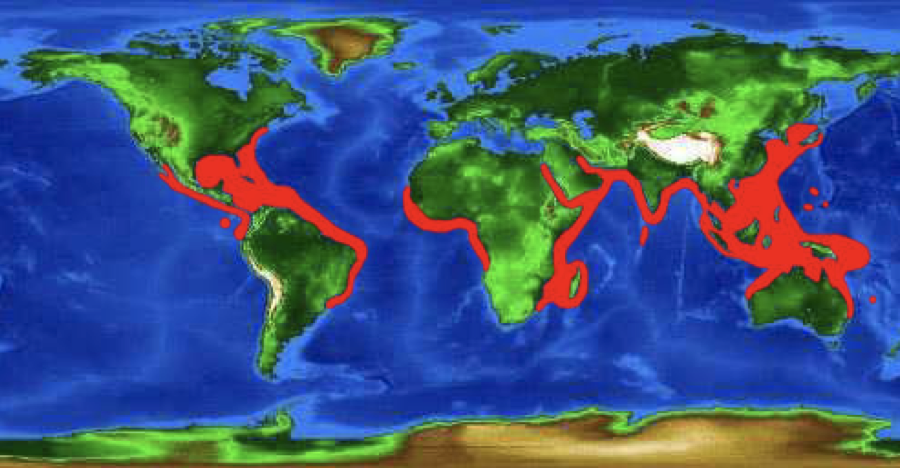
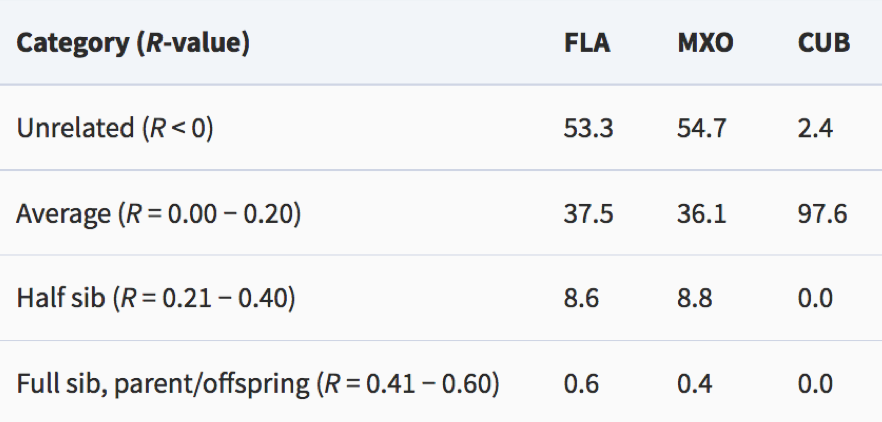
By Samantha Fisher The spotted eagle ray, Aetobatus narinari, are interesting marine organisms. There is little known about this ray species, but it is extremely important as not only does it play a role in the ocean’s food web, but many countries depend on A. narinari’s economic value. This paper gives an overview of the characteristic features of A. narinari, along with their diet, reproductive strategies, social structure, distribution, and predators. Also included is an overview of conservation concerns and the possibility of an A. narinari species complex. The spotted eagle ray is of the kingdom Animalia, phylum Chordata, class Chondrichthyes, order Myliobatiformes, and of the family Aetobatidae. It was scientifically described as Aetobatus narinari in 1790 by Euphrasen (Kyne et al., 2006). This ray is often referred to by a multitude of names in the English language including the bishop ray, lady ray, and the leopard ray (Bester), as well as the Bonnetray and Maylan among others (Kyne et al., 2006). A. narinari has a distinct set of characteristic features. They have a dorsoventrally flattened body (Morrissey et al., 2018), with their wingspan often doubling their body length. The tail of A. narinari can be up to three-times as long as the ray’s main body when unharmed and there are a number of barbs, ranging from two to six, present. The mouth is located on the ventral, or bottom, side of their body allowing for benthic feeding. These organisms have a unique dorsal coloration pattern, with small white dots and rings contrasting against a variation of black, gray, or brown body colors. The ventral portion of the body is usually white (see Figure 1). A. narinari can be up to five meters long including their tail. Their weight varies, with the largest recorded ray reaching two-hundred thirty kilograms (Bester). The diet of A. narinari includes a variety of mollusks, echinoderms, and osteichthyes. Because their mouth is located on the ventral portion of their body, they are also able to search through the benthos to find invertebrates. This ray does not have dentition similar to their shark and skate relatives. Instead, they have a series of grinding plates that allow the ray to crush and grind up their prey items. Because of their dentition, A. narinari is able to remove their prey item’s flesh from their shell using papillae in their mouth. This can be seen when the stomach contents of A. narinari are observed, as prey items are present in the stomach, but these prey items lack their shells. (Bester). The reproductive strategies of A. narinari are not well known. In fact, the female reproductive strategies have rarely been studied, as the collection of mature organisms is difficult and the preservation of collected organisms has not resulted in well preserved reproductive organs (Schluessel et al., 2010). Schluessel et al. (2010) noted that only the left ovary and uterus are functional, as the right uterus is smaller. They also noted that this pattern occurs in many related species. It is known that A. narinari are ovoviviparous, with the eggs developing and hatching within the mother and are then released. They have relatively small litters, with one to four rays being born per litter (Bester). A. narinari has a global distribution, with habitats in most temperate and tropical waters (see Figure 2). These rays are often distributed coastally, but they can be found in waters up to sixty meters deep. They can also be found near coral reefs and in some estuaries and lagoons. Although individuals are often seen near coastal regions, it is believed that it may be possible for them to transverse ocean basins (Kyne et al., 2006). These rays are known to have an increased rate of site fidelity, often inhabiting or returning to the same locations at multiple points in their lives (Bester). The social structure of A. narinari populations are also not well known. As Sellas et al. determined in their 2015 study, genetic diversity of populations may vary. There are some populations in Florida and Mexico where individuals are approximately fifty percent related, with populations in Cuba being less than three percent related (see Table 1). The number of family members per population vary around the world, with no two populations alike (Sellas et al., 2015). It has been noted that population sizes can range between a single individual to schools of hundreds of individuals (Kyne et al., 2006). There are also data supporting the seasonal migration of some ray populations, specifically populations off of the coast of Florida, that migrate seasonally, moving to warmer waters during the winter months. Like genetic diversity, the seasonal migration of populations varies. There are many populations that migrate seasonally and there are other populations that stay in same location year around (Sellas et al., 2015). Predators of A. narinari include many of species of shark. The sharks target individuals of any age, ranging from newborns to older rays. Parasites, worms, and leeches also target A. narinari, causing a variety of problems for infected individuals (Bester). While biological predators are important to note, the largest threat to A. narinari is humans. These rays are usually caught unintentionally as bycatch; however, there are some instances when A. narinari is considered the target species. There are artisanal fisheries for A. narinari in the southern portion of the Gulf of Mexico and in the northeastern portion of Venezuela. These unregulated fisheries do not target a specific age group, size, or coloration; they target all individuals (Tagliafico et al., 2012). According to Tagliafico et. al (2012), 55.9 metric tons of A. narinari were caught between 2005 and 2007 in Venezuela. A. narinari is also caught in many other countries such as most of Southeast Asia, Africa, and Australia. There are some countries in which these rays are protected. For example, they are protected in Florida, USA, in the Maldives, as export of ray products is against the law, and in the marine protected areas of Australia (Kyne et al., 2006). Conservation of A. narinari is becoming exceedingly important, as they are being targeted in multiple fisheries and there is limited knowledge about their lifestyle. According to the ICUN Red List of Threatened Species, A. narinari has been listed as “near-threatened” on a global scale since 2006 and is listed as “vulnerable” in Southeast Asia (Kyne et al., 2006). The conservation status of the species has not been assessed since 2006 and should be updated as soon as possible or as soon as new research is available. While A. narinari has historically been referred to as a single species, there is some compelling evidence that it may be a part of a species complex. Richards et al. (2009) were able to delineate 2 separate species of A. narinari using both mitochondrial and nuclear DNA, as well as multiple genetic approaches. They determined that there is a species specific to the western and central Pacific Ocean and a species specific to the central Atlantic Ocean and the eastern Pacific Ocean. The second species can be divided into 2 distinct subspecies separated by Panama (Richards et al., 2009). This research is extremely important to note, as it makes conservation of the A. narinari species complex imperative. Because of the species complex, there are now less known individuals of each species, causing an increased concern for species conservation. Although it is now known that a species complex exists, A. narinari is still referred to as a single species. In conclusion, the spotted eagle ray (A. narinari) is a species with a unique set of characteristics. The coloration pattern, incredible size, and global distribution, among other traits, all make A. narinari unique. Large numbers of these rays are targeted in unregulated artisanal fisheries, causing concern for conservation of this “near-threatened” species among conservationists and scientists alike. As new information is brought to light concerning the possibility of an A. narinari species complex, it is important to push for stronger conservation efforts to protect these organisms.
References Bester, C. (n.d.). Aetobatus narinari. Retrieved from https://www.floridamuseum.ufl.edu/fish/discover/species-profiles/aetobatus-narinari# Kyne, P.M., Ishihara, H., Dudley, S.F.J., & White, W.T. (2006). Aetobatus narinari. The ICUN Red List of Threatened Species. Retrieved from http://www.iucnredlist.org/details/39415/0 Morrissey, J.F., Sumich, J.L., & Pinkard-Meier, D.R. (2018) Introduction to the Biology of MArine Life (11th ed). Burlington, MA: Jones & Bartlett Learning Richards, V.P., Henning, M., Witzell, W., & Shivji, M.S. (2009). Species Delineation and Evolutionary History of the Globally Distributed Spotted Eagle Ray (Aetobatus narinari). Journal of Heredity, 100(3), 273-283. Sellas, A.B., Bassos-Hull, K., Pérez-Jiménez, J.C., Angulo-Valdés, J.A., Bernal, M.A., & Hueter, R.E. (2015). Population Structure and Seasonal Migration of the Spotted Eagle Ray, Aetobatus narinari. Journal of Heredity, 106(3), 266-275. Schluessel, V., Bennett, M.B., & Collin, S.P. (2010). Diet and Reproduction in the White- Spotted Eagle Ray Aetobatus narinari from Queensland, Australia and the Penghu Islands, Taiwan. Marine and Freshwater Research, 65(11), 1278-1289. Tagliafico, A., Rago, N., Rangel, S., & Mendoza, J. (2012). Exploitation and Reproduction of the Spotted Eagle Ray (Aetobatus narinari) in the Los Frailes Archipelago, Venezuela. Fishery Bulletin, 110(3), 307-316.
0 Comments
Leave a Reply. |
Biology of the Sea
|

 RSS Feed
RSS Feed
