|
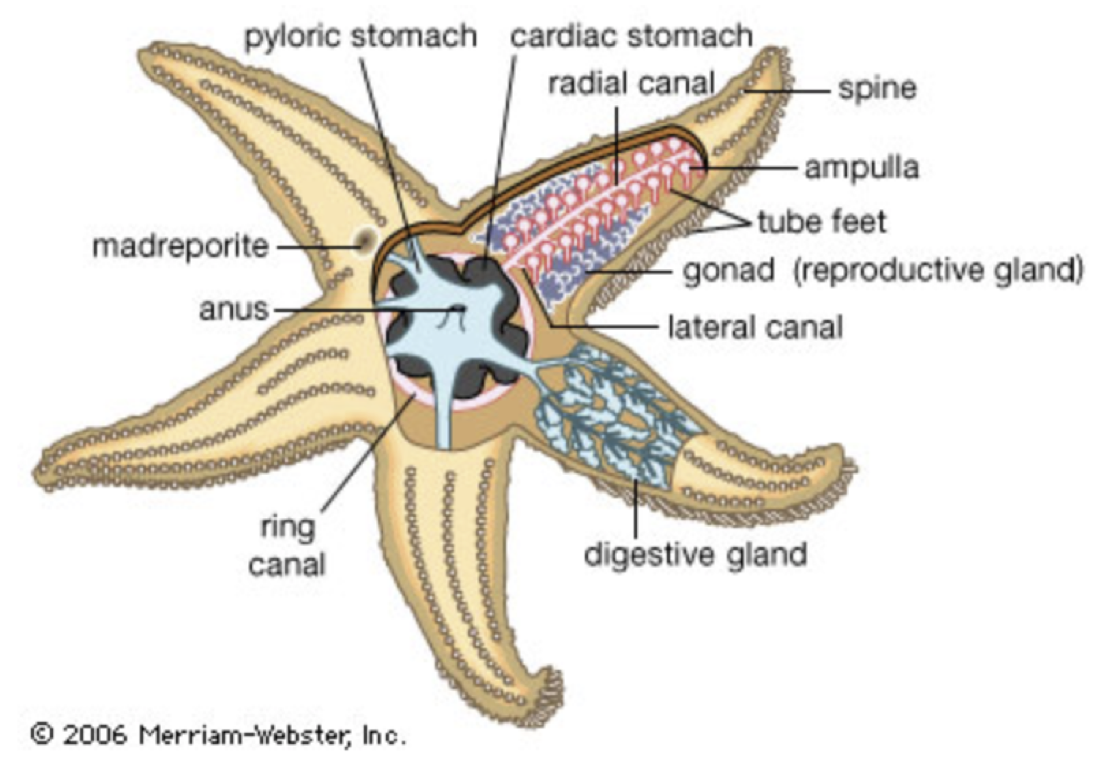
By Eleanor Winston Things are not always what they seem and when it comes to the Asteroidea, or the starfish, they are not the angelic peaceful creatures humans make them out to be. The starfish’s greatest weapon is its feet. They use them to sense their surroundings, eat and capture prey, for transportation, and even reproduction. Starfish are also known to be the “keystone” predators in most of their ecosystems which have kept other species from having outbreaks. However, various species have become detrimental to some coral reefs due to overpopulation. This research paper will explain and expose the starfish species in their daily lives. Asteroidea, or more commonly known as Starfish, are all a part of the phylum Echinodermata. Echinoderms consist of various species of Brittle Stars, Sea Cucumbers, Sea Urchins, and Crinoids. One major characteristic most Echinoderms have in common is that they live in intertidal and subtidal zones and are generally benthic creatures. Benthic meaning creatures that generally live on the ocean floor and do not swim around. Asteroidea generally live on the ocean floor, however, through research, scientists have observed that some starfish have been seen to float up to the surface of the water. (Echinoderm, 2008) The environment of a starfish ranges from soft floors such as mud and sand, to more rocky areas of the ocean. Scientists have estimated there are 2,000 species of starfish and all of them are saltwater organisms, meaning they cannot survive in freshwater. (Whale Facts) Usually, starfish generally live in places where food is easy to come by. This is why scientists may find starfish in giant packs where there is a common food source. (Echinoderms, 2008) The environment of a starfish is very flexible and different species are seen all over the world’s oceans. This is a result of their flexible physical structure, which ties in with the dangerous nature of the starfish. A starfish’s greatest weapon against the harsh environments of the ocean is its feet. Reproduction is arguably the most interesting way a starfish can use its feet. Most starfish reproduce through asexual reproduction and sexual reproduction. When a starfish reproduces sexually, it releases their egg and sperm through its feet into the water. These become fertilized embryos, which are categorized as zooplankton until they undergo metamorphosis. The starfish then settles down on the ocean floor, becoming an adult. The way in which starfish undergo asexual reproduction is through fragmentation. This is where a part of the starfish's limb detaches from its body and then forms into another starfish. (Echinoderms) After asexual reproduction, starfish can regenerate their lost limb and continue on with their lives. This is a great defense mechanism against predators and makes it much harder to kill them off. The ability to regenerate starts with the combination of morphallaxis cells and epimorphosis cells. Regeneration is similar to starfish growing an arm naturally, however it takes a lot of energy to regrow a foot. This limits the organism’s eating habits and requires them to use a lot less energy in their daily lives. (Lawrence, 2013) Most species of Asteroidea have five to forty limbs, which extend from their central body disc, or the center of the organism. Starfish use their limbs to navigate away from danger or towards prey, therefore they are not sessile creatures.(Starfish 2008) Sessile meaning creatures that cannot move on its own, such as a sponge. According to Philip’s encyclopedia, starfish move through their water vascular system. It begins by having water enter through the sieve plate where the water is drawn in by tiny hairs that travel through the five radial canals into their tubed feet. The ampulla will start to contract and water is forced into the foot which allows them to attach to hard rocks. Then, the muscles in the foot shorten which allows water to be forced back into the foot, and thus draw the starfish forward (figure 3). From these steps, it is obvious their methods of transportation are very slow, which is one of their greatest weaknesses. Another characteristic starfish possess is radial symmetry, which allows the organisms to be divided into almost identical halves in every angle of the central axis (Starfish 2008). Another known physical trait starfish are known for is that they do not have a brain. However, that does not make them dumb creatures, like they are portrayed through the famous character Patrick Star in the well-known kid’s TV show Spongebob Squarepants. Instead, they use sensory organs that makes them the perfect predators to bottom feeders. One of the sensory organs they possess is an eyespot, which helps to differentiate between light and dark and gives them the ability to locate any danger or even their next victims. A starfish captures and consumes their prey through grabbing it with their tubed feet. Typically, if the prey has a shell, they have the ability to crack it open, thus making the animal defenseless. Starfish have two stomachs located on the underside of their body. (figure 3) One allows them to capture and the other digests their prey. (figure 2). It can even extend one of its stomachs through its mouth and surround their prey. (Starfish, 2008) As for the food they eat, starfish are carnivores so most of their prey consists of mollusks, crustaceans, sea worms, and plankton. They also feed on bottom debris and most anything they can get their mouths around. This makes them very important species in the ecosystems they live in because they control outbursts of different herbivores. Starfish are the main predator for many intertidal and subtidal environments and have been found to be the keystone predator of the ecosystems they live in. Keystone predators are carnivores that help maintain outbreaks of other species. Though their main diet consists of species from the phylum Mollusca, the starfish is not very picky when it comes to what they consume. On the other hand, starfish can be detrimental to some environments like coral reefs. In 1963, a population of starfish called Acanthaster planci (figure 1) exploded on the great barrier reef. The outbreak of this species killed off a lot of coral and fisherman targeted to kill off this species. Since the first outbreak in Australia, there have been more outbreaks in the southern Pacific. Scientists have yet to understand why these outbreaks happen, but the major hypothesis blames the endangerment of the ‘Pacific Triton,’ the starfish’s biggest predator (Crown of Thorns Starfish, 2017). Starfish are not endangered species but they do have threats just like any other organism. Due to their small size and inability to move fast or defend themselves, they are easy targets to bigger predators such as sharks, various species of crabs, and even humans. Due to their slow nature, they cannot always move fast enough to get back into the water. If the tide does not bring them back, they will become stranded on the shore, drying out and dying, becoming food for birds on the land. (Whale Facts) Starfish are beautiful, yet ruthless organisms. The way they eat, regrow their arms, transport, and capture their prey are just a few interesting characteristics of the species that make them dangerous to other benthic creature. They can live in many diverse ecosystems in which they are the keystone predators, which are essential to the survival of the food chain. On the other hand, starfish have been known to ruthlessly destroy ecosystems like the Great Barrier Reef. Starfish are not endangered mostly because they have a very good defense system. However, due to their inability to move quickly, it can make them defenseless when they are washed up on shore or faced with bigger predators. Overall, starfish are very important to the ecosystem despite their ruthless nature when it comes to coral reefs. It is important society needs to protect this creature, or else many ecosystems will be lost.
Works Cited Starfish. (2008). In Philip's encyclopedia. London, UK: Philip's. Retrieved from https://login.liblink.uncw.edu/login?url=http://search.credoreference.com/content/entry/philipency/starfish/0?institutionId=3329 Crown-of-thorns starfish. (2017). In Encyclopaedia Britannica, Britannica concise encyclopedia. Chicago, IL: Britannica Digital Learning. Retrieved from https://login.liblink.uncw.edu/login?url=http://search.credoreference.com/content/entry/ebconcise/crown_of_thorns_starfish/0?institutionId=3329 Echinoderms. (2008). In C. Love, & C. Stamps (Eds.), Animals: a visual encyclopedia. London, UK: Dorling Kindersley Publishing, Inc. Retrieved from https://login.liblink.uncw.tedu/login?url=http://search.credoreference.com/content/entry/dkanimals/echinoderms/0?institutionId=3329 Echinoderms. (n.d.). Retrieved October 25, 2017, from http://www.mesa.edu.au/echinoderms/echino01.asp Starfish. (n.d.). Retrieved October 25, 2017, from http://www.whalefacts.org/starfish-facts/ Lawrence, J. M. (2013). Starfish: biology and ecology of the Asteroidea. Starfish. (2017). In Encyclopaedia Britannica, Britannica concise encyclopedia. Chicago, IL: Britannica Digital Learning. Retrieved from https://login.liblink.uncw.edu/login?url=http://search.credoreference.com/content/entry/ebconcise/starfish/0?institutionId=3329
0 Comments
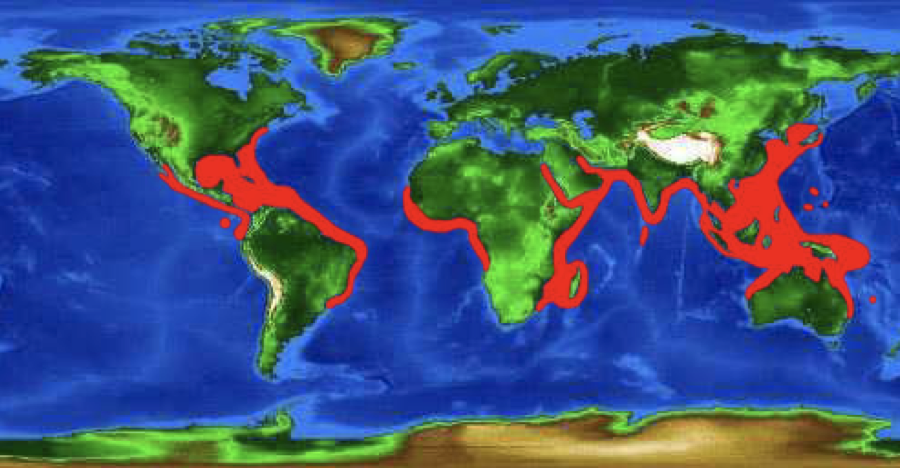
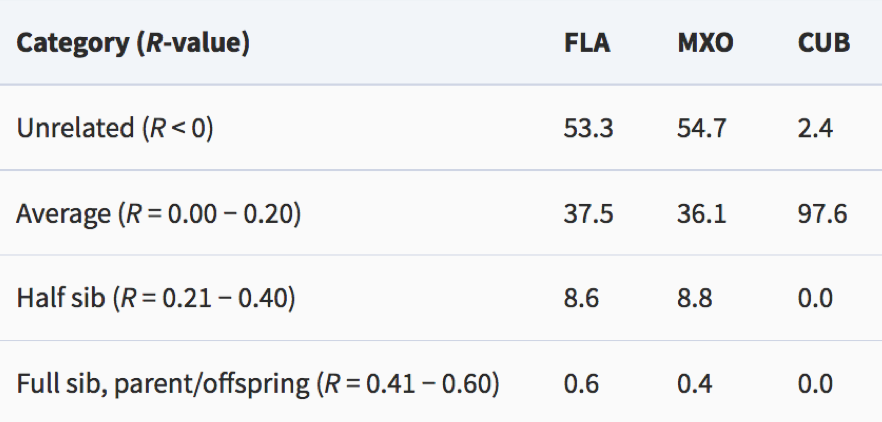
By Samantha Fisher The spotted eagle ray, Aetobatus narinari, are interesting marine organisms. There is little known about this ray species, but it is extremely important as not only does it play a role in the ocean’s food web, but many countries depend on A. narinari’s economic value. This paper gives an overview of the characteristic features of A. narinari, along with their diet, reproductive strategies, social structure, distribution, and predators. Also included is an overview of conservation concerns and the possibility of an A. narinari species complex. The spotted eagle ray is of the kingdom Animalia, phylum Chordata, class Chondrichthyes, order Myliobatiformes, and of the family Aetobatidae. It was scientifically described as Aetobatus narinari in 1790 by Euphrasen (Kyne et al., 2006). This ray is often referred to by a multitude of names in the English language including the bishop ray, lady ray, and the leopard ray (Bester), as well as the Bonnetray and Maylan among others (Kyne et al., 2006). A. narinari has a distinct set of characteristic features. They have a dorsoventrally flattened body (Morrissey et al., 2018), with their wingspan often doubling their body length. The tail of A. narinari can be up to three-times as long as the ray’s main body when unharmed and there are a number of barbs, ranging from two to six, present. The mouth is located on the ventral, or bottom, side of their body allowing for benthic feeding. These organisms have a unique dorsal coloration pattern, with small white dots and rings contrasting against a variation of black, gray, or brown body colors. The ventral portion of the body is usually white (see Figure 1). A. narinari can be up to five meters long including their tail. Their weight varies, with the largest recorded ray reaching two-hundred thirty kilograms (Bester). The diet of A. narinari includes a variety of mollusks, echinoderms, and osteichthyes. Because their mouth is located on the ventral portion of their body, they are also able to search through the benthos to find invertebrates. This ray does not have dentition similar to their shark and skate relatives. Instead, they have a series of grinding plates that allow the ray to crush and grind up their prey items. Because of their dentition, A. narinari is able to remove their prey item’s flesh from their shell using papillae in their mouth. This can be seen when the stomach contents of A. narinari are observed, as prey items are present in the stomach, but these prey items lack their shells. (Bester). The reproductive strategies of A. narinari are not well known. In fact, the female reproductive strategies have rarely been studied, as the collection of mature organisms is difficult and the preservation of collected organisms has not resulted in well preserved reproductive organs (Schluessel et al., 2010). Schluessel et al. (2010) noted that only the left ovary and uterus are functional, as the right uterus is smaller. They also noted that this pattern occurs in many related species. It is known that A. narinari are ovoviviparous, with the eggs developing and hatching within the mother and are then released. They have relatively small litters, with one to four rays being born per litter (Bester). A. narinari has a global distribution, with habitats in most temperate and tropical waters (see Figure 2). These rays are often distributed coastally, but they can be found in waters up to sixty meters deep. They can also be found near coral reefs and in some estuaries and lagoons. Although individuals are often seen near coastal regions, it is believed that it may be possible for them to transverse ocean basins (Kyne et al., 2006). These rays are known to have an increased rate of site fidelity, often inhabiting or returning to the same locations at multiple points in their lives (Bester). The social structure of A. narinari populations are also not well known. As Sellas et al. determined in their 2015 study, genetic diversity of populations may vary. There are some populations in Florida and Mexico where individuals are approximately fifty percent related, with populations in Cuba being less than three percent related (see Table 1). The number of family members per population vary around the world, with no two populations alike (Sellas et al., 2015). It has been noted that population sizes can range between a single individual to schools of hundreds of individuals (Kyne et al., 2006). There are also data supporting the seasonal migration of some ray populations, specifically populations off of the coast of Florida, that migrate seasonally, moving to warmer waters during the winter months. Like genetic diversity, the seasonal migration of populations varies. There are many populations that migrate seasonally and there are other populations that stay in same location year around (Sellas et al., 2015). Predators of A. narinari include many of species of shark. The sharks target individuals of any age, ranging from newborns to older rays. Parasites, worms, and leeches also target A. narinari, causing a variety of problems for infected individuals (Bester). While biological predators are important to note, the largest threat to A. narinari is humans. These rays are usually caught unintentionally as bycatch; however, there are some instances when A. narinari is considered the target species. There are artisanal fisheries for A. narinari in the southern portion of the Gulf of Mexico and in the northeastern portion of Venezuela. These unregulated fisheries do not target a specific age group, size, or coloration; they target all individuals (Tagliafico et al., 2012). According to Tagliafico et. al (2012), 55.9 metric tons of A. narinari were caught between 2005 and 2007 in Venezuela. A. narinari is also caught in many other countries such as most of Southeast Asia, Africa, and Australia. There are some countries in which these rays are protected. For example, they are protected in Florida, USA, in the Maldives, as export of ray products is against the law, and in the marine protected areas of Australia (Kyne et al., 2006). Conservation of A. narinari is becoming exceedingly important, as they are being targeted in multiple fisheries and there is limited knowledge about their lifestyle. According to the ICUN Red List of Threatened Species, A. narinari has been listed as “near-threatened” on a global scale since 2006 and is listed as “vulnerable” in Southeast Asia (Kyne et al., 2006). The conservation status of the species has not been assessed since 2006 and should be updated as soon as possible or as soon as new research is available. While A. narinari has historically been referred to as a single species, there is some compelling evidence that it may be a part of a species complex. Richards et al. (2009) were able to delineate 2 separate species of A. narinari using both mitochondrial and nuclear DNA, as well as multiple genetic approaches. They determined that there is a species specific to the western and central Pacific Ocean and a species specific to the central Atlantic Ocean and the eastern Pacific Ocean. The second species can be divided into 2 distinct subspecies separated by Panama (Richards et al., 2009). This research is extremely important to note, as it makes conservation of the A. narinari species complex imperative. Because of the species complex, there are now less known individuals of each species, causing an increased concern for species conservation. Although it is now known that a species complex exists, A. narinari is still referred to as a single species. In conclusion, the spotted eagle ray (A. narinari) is a species with a unique set of characteristics. The coloration pattern, incredible size, and global distribution, among other traits, all make A. narinari unique. Large numbers of these rays are targeted in unregulated artisanal fisheries, causing concern for conservation of this “near-threatened” species among conservationists and scientists alike. As new information is brought to light concerning the possibility of an A. narinari species complex, it is important to push for stronger conservation efforts to protect these organisms.
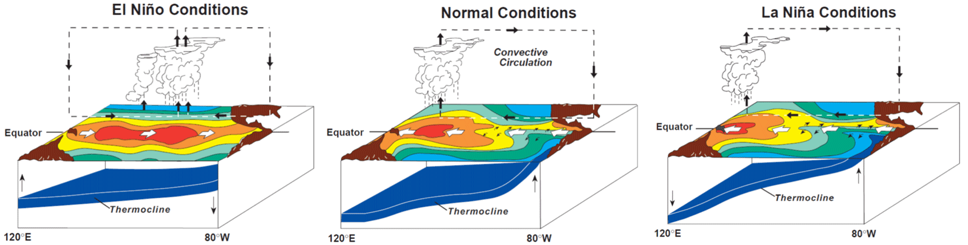
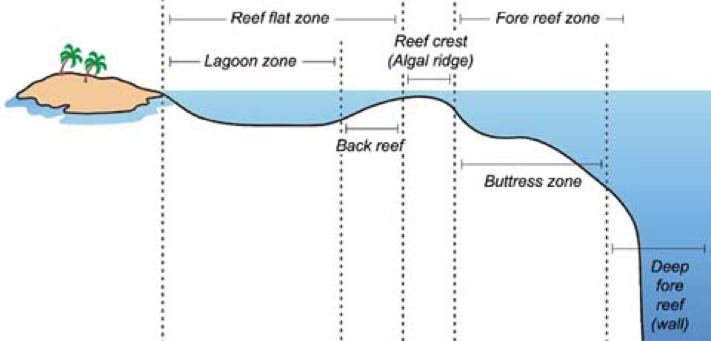
References Bester, C. (n.d.). Aetobatus narinari. Retrieved from https://www.floridamuseum.ufl.edu/fish/discover/species-profiles/aetobatus-narinari# Kyne, P.M., Ishihara, H., Dudley, S.F.J., & White, W.T. (2006). Aetobatus narinari. The ICUN Red List of Threatened Species. Retrieved from http://www.iucnredlist.org/details/39415/0 Morrissey, J.F., Sumich, J.L., & Pinkard-Meier, D.R. (2018) Introduction to the Biology of MArine Life (11th ed). Burlington, MA: Jones & Bartlett Learning Richards, V.P., Henning, M., Witzell, W., & Shivji, M.S. (2009). Species Delineation and Evolutionary History of the Globally Distributed Spotted Eagle Ray (Aetobatus narinari). Journal of Heredity, 100(3), 273-283. Sellas, A.B., Bassos-Hull, K., Pérez-Jiménez, J.C., Angulo-Valdés, J.A., Bernal, M.A., & Hueter, R.E. (2015). Population Structure and Seasonal Migration of the Spotted Eagle Ray, Aetobatus narinari. Journal of Heredity, 106(3), 266-275. Schluessel, V., Bennett, M.B., & Collin, S.P. (2010). Diet and Reproduction in the White- Spotted Eagle Ray Aetobatus narinari from Queensland, Australia and the Penghu Islands, Taiwan. Marine and Freshwater Research, 65(11), 1278-1289. Tagliafico, A., Rago, N., Rangel, S., & Mendoza, J. (2012). Exploitation and Reproduction of the Spotted Eagle Ray (Aetobatus narinari) in the Los Frailes Archipelago, Venezuela. Fishery Bulletin, 110(3), 307-316. By Balma Sales Diaz INTRODUCTION The Great Barrier Reef is considered one of the seven wonders of the natural world and is considered the largest living organism on Earth. It is located in Australia. Its amazingly vivid corals and thousands of animals inhabiting it make up the beauty of this reef, making it one of the most touristic locations in the world. However, it will not last much longer if humans don’t act to stop the terminal stage the Great Barrier Reef is facing (Hoegh-Guldberg et al., 2009). This paper will discuss firstly the idiosyncrasy that forms Coral Reefs, and will continue by going into more detail about the Great Barrier Reef and what environmental causes have led it to a difficult terminal stage to recover from. CORAL REEFS Coral reefs are seen as immensely vast animals whose extension can reach hundreds of kilometers and provide habitat to a large number of organisms in tropical and subtropical areas. They are composed of coral polyps, which are tiny animals that produce a carbonate infrastructure for the reef where these organisms live. Their growth is affected by many factors such as light intensity, length, water temperature, plankton concentrations, and the deposition and removal of calcium carbonate (CaCO3); therefore, they are merely found in salinity warm seawaters where reefs can be built. On the other hand, corals’ light absorption occurs through zooxanthellae, which is a photosynthetic alga that lives in the corals tissue and provides them with a mutualistic symbiotic relationship. Turbidity can also affect the growth of corals depending on the levels of suspended sediment in the water column. The higher the levels, the lower the existence of reefs. Coral reefs, which reproduce asexually or sexually, compose the largest living creatures on Earth and even though there are many around the globe, the most popular one is located in Australia (Morrissey et al., 2018). THE GREAT BARRIER REEF (GBR) The Great Barrier Reef is an oceanic reef classified as the largest coral reef ecosystem in the world and the only living organism that can be seen on Earth from space (see Figure 1). It covers 360,000 square km, and the coral reefs stretch up to 2,575 km. Its waters can go over 2,000 meters deep and it encompasses 2,500 individual reefs. There are 400 different types of corals, 1,500 different fish species, 4,000 types of mollusk, and 240 different species of seabirds and many other organisms (UNESCO, 2017). Origin and Evolution The GBR found in Queensland’s continental shelf did not form until 6,000 years ago which is a relatively short period of time considering Earth’s history. During glacial and interglacial cycles, the area has been exposed to extreme conditions of sea levels rising and dropping which have enhanced the growth of these corals. Moreover, reefs are built mainly when sea levels rise indicating that the GBR was created during interglacial periods (Hoegh-Guldberg et al., 2009). Geomorphology The GBR is composed of three zones of reefs that go from fringing reefs to mid-shelf reefs and outer shelf reefs. All of them have major energy conditions in which the stronger these conditions are, the more different their zonation. Normally, coral reefs have a protectionist coral reef front which is steeper in high energy areas (see Figure 2). Afterwards, the reef crest appears and we can find a diversity of living organisms below the low spring tide area. It is then followed by a reef flat that contains the highest part of the reef and aligned, the densest area with coral colonies growing. At the back, there is the most vulnerable coral colonies growing in a slope of sand. Moreover, it is also important to highlight that the GBR does not only contain reefs and other organisms, but also some coral islands produced by the accumulation of sediments on the reef flat due to the action of waves. They can be classified by types of sediments and vegetation cover which includes from the unvegetated to highly abundant vegetation. The latter are important because they have different distribution patters. Vegetated islands exist on the far north and south but are absent on the central part of the Great Barrier Reef; nonetheless, unvegetated islands can be found along the entire GBR being less frequent on the center (Hoegh-Guldberg et al., 2009). HABITAT & ECOSYSTEM The GBR rises in a shallow continental shelf with a 75 km wide stretch of water located at the center, called GBR Lagoon where many reefs inhabit. This Lagoon absorbs nutrients and solids that are carried inshore after heavy rainfalls due to the force of rivers situated along the coast. At the Coastal Sea there are oceanic tides that also heavily influence some parts of the Great Barrier creating tidal nodes that end up forming a large number of tides in the area and strong currents. Along with sediment grain size, turbidity, depth and nutrients, all of these previously defined factors influence the habitat of coral reefs. Moreover, there are two major environments found in the Great Barrier Reef: the most common which represents 50% of the seabed and where we can find coarse sands, algae and benthos; and the less but more complex environment which is found in relatively less parts. Physical Factors Influencing the Ecosystem Mud is one of the most important factors found which conforms a great amount of the GBR’s length. Worms, shrimps and bivalves inhabit this inshore area even though there are still a few mobile animals which are included too. Offshore where turbidity is lower, muddy territory has the same fauna as inshore with a little bit more diversity. Moreover, muddy areas that are located further away from the shelf and which are quite sandy, support a wide range of surface biota such as starfish, crabs, fish and algae while supporting many bio habitats. On the other extreme there is Benthic Stress (BS) which is the cause of the largest tidal range in the GBR, provoking harsh currents that push sediments away to leave stones and rocks exposable to anything. As muddy territories, benthic stress also provides habitat to a large number of highly diversified fauna and moderated biomass. Upwellings play an important role as a physical factor when providing nutrients in deep clear waters. These areas have a moderate diversified fauna and biomass, and are located offshore out of the range of muddy and turbid areas, where light absorption is easier to reach the seabed (Hoegh-Guldberg et al., 2009). Development of Coral Reefs Corals in the GBR are supported by carbonated reefs and rock bases that geologically evolved over time. Fixing these carbonates to make them hard substrates in marine systems is a very important geological role for them (Morrissey et al., 2018). Besides carbonate ions, other factors affect the development of coral reefs: temperature and light availability. Firstly, temperature variations such as El Niño (or la Niña) cycle which can produce extreme hot and cold temperatures, can influence the stress coral reefs have to deal with (see Figure 3). On the other hand, light dependence is a major determinant to where coral reefs are located in order to obtain high rates of photosynthesis. Water columns sometimes block light absorption because of heavy particles they contain, and are the main reason why coral reefs are not located next to major rivers. Changes temporally can also influence light like tidal variations or periods of cloudiness over the water (Hoegh-Guldberg et al., 2009). Primary Productivity The GBR was defined by Charles Darwin as a “marine rainforest existing in an oceanic desert” due to the fact that coral reefs have a very productive ecosystem, but the ocean that surrounds them does not. Coral reefs have a manifestly efficient mechanism to photosynthesize and recycle nutrients provided by organisms such as blue green bacteria (Cyanophyta), seaweeds, phytoplankton and dinoflagellates. Their productivity depends on whether they’re found inshore or offshore, in a high or a low latitude and the location within the reef. Moreover, primary production is not only based on sunlight and CO2, but also on inorganic nutrients such as inorganic nitrogen and phosphorus. Their job is to breakdown waste of animals which have consumer other organisms and debris generated by predators. This nitrogen is supplied to coral reefs through a nitrogen fixation process which is quite important to coral reefs. As previously stated, there are consumers and producers in coral reefs; however, more producers currently exist than consumers do, consequently because of high inefficiencies in nutrients and energy transfer in trophic interactions. Coral reefs trophic levels range from primary producers at the bottom to herbivores, predators and apex predators at the top. Among them, some organisms have mutualistic symbiotic relationships with coral reefs which is a hallmark of coral reefs in the GBR. These have been a result of the low nutrient conditions that rule the advantages in the primary producer-consumer relationship. An example of these distinctive relationships are reef-building corals and dinoflagellate protists. The latter provide energy in large amounts to the coral which, in return, deliver access to inorganic nutrients. As a result, photosynthetic rates skyrocket and strengthen the process of calcification (Hoegh-Guldberg et al., 2009). Biodiversity: More than fish The Great Barrier Reef is commonly popular because of its status as the largest living organism on Earth, but also because of the richness of its biodiversity. Biota is highly diverse due to the diversity of habitat found within coral reefs that can sway from soft sediments to pelagic and coastal habitats. Furthermore, the highest number of organisms is found in the so-called “coral reef triangle” which is in line with Indonesia and the Philippines, even though these numbers are declining due to the major threat the GBR is suffering nowadays which will be discussed in a later section (Hoegh-Guldberg et al., 2009). Onwards, the Great Barrier Reef is home to a large extent of animals that range from the tiniest plankton to hosting one of the most important dungong populations in the world besides the overly popular fish. Some of these animals are the following:
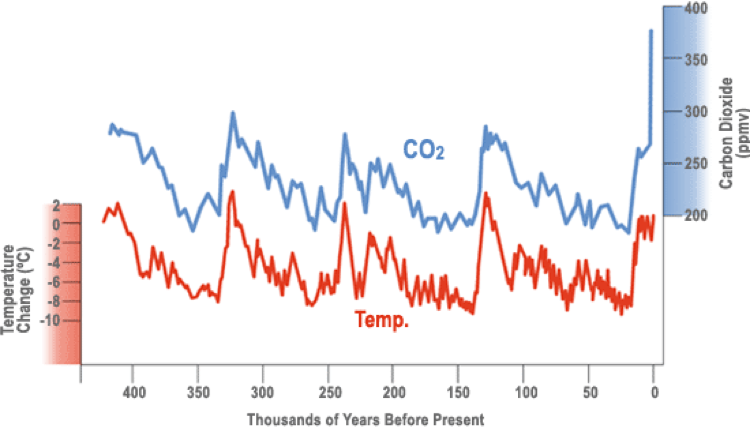
THREATS TO THE GBR In the past 30 years the Great Barrier Reef ecosystem has been threatened by different factors that are not only limiting its growth, but destroying it slowly. Coral reefs are strong and have always faced disturbances to which they have adapted to survive. However, now those disturbances are damaging more than ever and action has to be taken (Hoegh-Guldberg et al., 2009). Human Impact The interaction of humans with the ecosystem of the Great Barrier Reef has greatly influenced the terminal stage the great wonder confronts these days. Even though humans play a significant role on the regenerative processes of coral reefs, their impact causes more harm than good. Overfishing has become a trend since there has been an increase in demand for seafood and more fisheries are being built. As a consequence, many species have been reduced, dangerously affecting the food web of the GBR and No Take Areas (NTA) where it is prohibited to fish, are increasing. It is important to highlight that due to this overfishing, dugongs have declined by 90% in the previous years (Australian Government, 2017). The use of land due to agriculture, urbanization and other disturbances have caused soil erosion and great amounts of sediments that flow into rivers to be taken to the GBR accumulating there and damaging coral reefs (Hoegh-Guldberg et al., 2009). This land run-off is the cause of poorer water quality that has increased the amount of sediments and pesticides in the marine water (Australian Government, 2017). Moreover, coral harvesting for personal pleasure, and human recreational activities such as fishing, also contribute to the damage when conducted excessively (Hoegh-Guldberg et al., 2009). However, one of the most important and popular human impacts that is affecting the environment worldwide too is climate change. Climate Change The increase of greenhouse emissions produced by deforestation and burning of fossil fuels by humans has greatly increased the earth’s overall temperature over thousands of years (see Graph 1). However, it is greatly impacting coral death in the GBR. Climate change has led to coral bleaching which is a phenomenon where corals feel stressed and they expel the zooxanthellae with which they had a mutualistic relationship (Anonymous, 2015). Corals change from brown to white as a result of the bleaching which is caused by rising sea level temperatures due to climate change. (Morrissey et al., 2018). Extreme weather conditions such as floods and cyclones are consequences of climate change too. Six cyclones of the highest category, number 5, have affected the GBR in the past decade (Anonymous, 2015). Increase in the turbidity and reductions on the levels of salinity can kill corals or create imbalances in the ecosystem due to over productivity caused by the increase of nutrients. (Australian Government, 2017) Greenhouse emissions of CO2 are expected to increase up to high levels that will exceed the conditions to which corals have always overcome; however, some researchers have found that some corals have symbiont relationships which can handle great thermal tolerance; hence, this can increase the probabilities resisting climate change if they adapt and create new symbiont relationships with new zooxanthella’s. (Morrissey et al., 2018). As a consequence of coral bleaching, pathogenic bacteria are increasing coral diseases, and although these diseases affect a low percentage of corals, it is still something to take care of (Hoegh-Guldberg et al., 2009). Ocean Acidification Out of all the impacts recorded and damaging the GBR, ocean acidification is the most significant one. Half of the CO2 that enters the atmosphere is absorbed by the ocean, and almost half of the CO2 created by humans (anthropogenic CO2) is found on 10% of the upper levels of the ocean. (Gangjian, et al., 2009). The main issue with ocean acidification is the damage of calcification rates in marine organisms. Its increase has produced large reductions on the Ph of the ocean, which produces calcification therefore increasing coral mortality rates (Gangjian et al., 2009). Coral reef building and habitat creation do not need high levels of acidification to be strongly affected; hence, the ongoing increase of CO2 levels in the water, can completely obliterate the GBR fauna and flora if greenhouse emissions are not reduced. (Australian Government, 2017). Path To Sustainability There are many reasons why the Great Barrier Reef needs immediate protection and action from everyone. Consequently, in 1975 the Great Barrier Reef Marine Park (GBRMP) was created to stabilize and protect it to ensure sustainability. This Marine Park was established in the coast of Queensland and it works cooperatively with the Australian government (Hoegh-Guldberg et al., 2009). The GBR is not only important because of the many species that provide such wonderful marine diversity in the largest coral reef ecosystem, but also because of the value and economic benefits it provides to Australia. It provides thousands of jobs for people and is one of the most visited touristic attractions of the world. Almost 2 million tourists visit it every year contributing on 90% of the economic activity of the large coral barrier reef (Australian Government, 2017). It can also influence human well-being of the community that relies upon it. Its beauty and the connection that many families living in Queensland have shaped over time with the natural wonder, contribute to a healthier environment. The Reef is not only a food source for numerous people, but a place to relax and enjoy the natural world that surrounds as well. In addition, it protects residents in the coast from extreme weather conditions that can result in strong wave actions (Australian Government, 2017). SUMMARY The Great Barrier Reef is the largest coral reef ecosystem in the world containing thousands of different species that make it unique. The Reef has always been faced with many disturbances from glacial to interglacial periods to which it has adapted in order to evolve and survive. However, in the last 30 years it seems that the reef’s adaptation to the extreme conditions that affect it nowadays is considerably challenging. Human impact has prompted the increase of climate change, consequently affecting ocean acidification and coral bleaching, which are the two main sources that are significantly damaging coral reef building and increasing coral mortality. Nevertheless, other more direct activities such as illegal fishing or water pollution from land based run-offs are critically impacting the ecosystem as well. The simplest solution to this problem is the reduction of CO2 absorbed in by the ocean. In order to implement it, international human interaction is essential. The Australian Government established the GBRMP to protect the Reef and sustain it to ensure its survival. The Marine Park can undertake as many actions as they’re capable of, but without the help of global cooperation to fight against global warming, these actions will be useless in the long-run. It is important to make people understand that if we want the future to survive climate change, first we have to start by saving the Earth and that implies the nature that makes us alive.
REFERENCE LIST
Anonymous. (2015). Judgement Day; Australia’s Great Barrier Reef. The Economist, 415, 38. Australian Government (2017, n.d). Australian Government: Great Barrier Reef Marine Park Authority. Retrieved from http://www.gbrmpa.gov.au Gangjian, W., Graham, M., Luhua, X., Malcolm, T. M., & Wengfeng, D. (2009). Evidence for Ocean Acidification in the Great Barrier Reef of Australia. Geochimica et Cosmochimica Acta, 73, 2332-2346. Hoegh-Guldberg, O., Hutchings, P., & Kingsford, M. (2009). The Great Barrier Reef: Biology, Environment & Management. Australia, New Zealand: CSIRO Publishing. Morrissey, J. F., Pinkard-Meier, D. R., & Sumich, J. L. (2018). Introduction to the Biology of Marine Life. Burlington, MA: David D. Cella. UNESCO (2017, n.d). UNESCO: World Heritage Convention. Great Barrier Reef. Retrieved from http://whc.unesco.org By Emilee Hawks The Florida Reef is the only living coral reef in the continental U.S. It is also the third largest coral reef in the world. Throughout this paper I will be explaining the various details about the Florida Reef ranging from the basic facts about its coral reef, to the marine animals that inhabit it, why the Florida coral reef is crucial to marine and human lives, and the dangers that affect the coral reefs considerably. Florida’s Coral Reef came into the world around 10,000 years ago trailing the last Ice Age. This happened when the sea’s water levels rose and revealed a growing coral reef. Coral reefs are in a constant state of fluctuation, where new living tissue grows on the outer surface. The skeleton from previous coral erodes to help make the sand that we find on our beaches today. The Florida Reef is over 150 miles long and 4 miles wide. The Florida Reef Tract stretches approximately 360 linear miles from Dry Tortugas National Park, west of the Florida Keys to the St. Lucie Inlet in Martin County (FDep, 2017). Growth of coral reef is a very strenuous process for it is relatively slow. Each coral reef is broken up into colonies, and those colonies grow only ½ inch to 7 inches a year. Although the growth is leisurely, Florida’s coral reef is home to specialized habitats that provide shelter, food, and breeding sites for numerous plants and animals such as; the spiny lobster, snapper, and grouper (Lapointe, 2004). Along with being a safe space for numerous animals and plants, Florida’s coral reef also stands as the foundation of a dynamic ecosystem with tremendous biodiversity (Beach, 2006). Every coral reef is important, but there are distinct reasons to why the Florida coral reefs are an essential segment of the sea’s ecosystem. The Florida reef tract is comprised of several outer reefs, and patch reefs, all of which hold over fifty species of coral. Located solely on the Florida reef tract resides over 80% of all coral reef species in the Tropical Western Atlantic, and it also houses over one hundred different species of fish (Leopold, 2001). Florida coral reefs are also indispensible for human lives as well. Having a diverse number of fish in the Florida reef tract makes commercial fishing for spiny lobster and stone crab the largest in the state (Pawlik, 2014). Being exposed to long periods of prosperous environments, the Florida coral reef has grown to awe-inspiring heights and variety making it a go-to tourism location. The Florida Keys hosts several million divers, snorkelers, boaters, and fishermen annually. A tremendous amount of people have became flabbergasted by the beauty of the coral reefs, so enthralled that they made Florida the number one dive destination in the world (Leopold, 2001). As stated before, the Florida coral reef is home to a remarkable amount of fish. There are certain species of fish that only inhabit Florida’s coral reef. These fish populate the coral reef for the reef provides shelters from predators, and they also provide a protected place for their breeding grounds. For example, the Florida reef is home to the; Banned Butterflyfish, Bluestripped Grunt, Blue Tang (or as I like to call them Dory Fish), French Angelfish, Porcupinefish, Queen Angelfish, Redband Parrotfish, Sargeant Major, Spotted Drum, Trumpetfish, and the Yellowtail Snapper. These beautiful fish dwell in the sanctuary the Florida coral reef provides. With the beauty that the Florida Reef provides, there are also endangerments that are acquired. Florida coral reefs are very, very delicate. Even the slightest touch can crush the fragile living coral polyps on the surface of each coral formation, exposing the entire coral head to infection and disease (Beach, 2006). Although the coral reefs bring in divers and snorkelers, the sightseers that touch, stand, or scrape the coral with fins, hands, or equipment damage that has grown for hundreds of years (Leopold, 2001). These are just examples of some of the travellers can cause; there are other humane damages that affect the reef. As a population we often sail out to sea to find food and other adventures, but with our boats comes damages that are likely to happen to the reef. Large boats often travel over Florida’s coral reef, and with that centuries of coral growth can be broken into irreplaceable fragments upon the first moment of collision with the vessel (Lapointe, 2004). Dredging is also an issue with damaging the reef. Dredging is when a boat enters shallow water and their propellers stir up the bottom of the water. In dredging’s wake leaves a milky white trail of calcium carbonate silt. This silt blocks the sunlight from reaching the coral reefs, and it can also asphyxiate the bottom-dwelling organisms (Leopold, 2001). Oil population is another big factor in damaging the reef. Oil pollution can happen from bilge discharge, untreated sewage from cruises, and increased turbidity from boat wakes reduce the conditions required for healthy coral growth. The hydrocarbons from oil pollution also pollute the water column and are absorbed into food chain in the organisms that the coral reef benefits from (Leopold, 2001). In conclusion the Florida reef is a spectacular place in which marvelous events happen. Not only are the sight from the coral reef breath taking, there are also multiple benefits for marine and human life that originates from the coral reef. Whether it be having a place for the Florida population to catch fish, or even just to serve as a vacation destination, the Florida reef is an amazing creation that should be protected and helped to strive. Works Cited
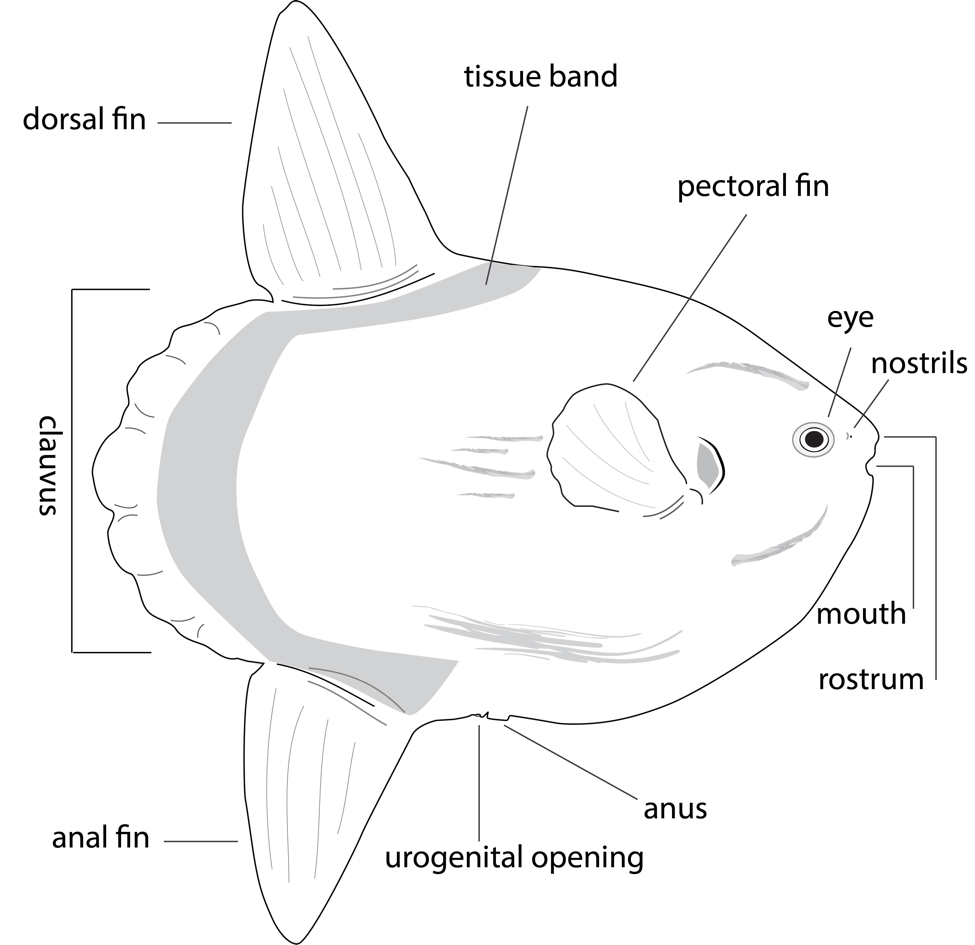
Deignan, L. K., & Pawlik, J. R. (2014). Perilous proximity: Does the Janzen–Connell hypothesis explain the distribution of giant barrel sponges on a Florida coral reef? Coral Reefs,34(2), 561-567. doi:10.1007/s00338-014-1255-x Beach, K. S., Walters, L. J., & Borgeas, H. B. (2006). Irradiance and nutrient limitation of Dicytota spp. populations on Conch Reef, Florida Keys, USA. Journal of Experimental Marine Biology and Ecology,329(1), 101-112. doi:10.1016/j.jembe.2005.08.010 Lapointe, B. E., Barile, P. J., & Matzie, W. R. (2004). Anthropogenic nutrient enrichment of seagrass and coral reef communities in the Lower Florida Keys: discrimination of local versus regional nitrogen sources. Journal of Experimental Marine Biology and Ecology,308(1), 23-58. doi:10.1016/j.jembe.2004.01.019 Where is Florida's coral reef? (n.d.). Retrieved October 28, 2017, from http://reefrelieffounders.com/florida-keys-reefs.html Florida's Coral Reefs. (n.d.). Retrieved October 28, 2017, from https://floridadep.gov/fco/fco/content/floridas-coral-reefs By Michael Austin Coward Just as the ocean is vast, so is the incomprehension of many of the organisms living in it. One such organism that very little is known about is the Mola mola. Also known as the ocean sunfish, the Mola mola is a mysterious, oddly shaped, pelagic fish that is difficult to photograph and research due to its skittish nature and deep ocean distribution (Cardone, 1992, p. 127). The Mola is known as the world’s heaviest bony fish (Shapiro, 2013). The research on this unique fish has expanded in recent years, led in part by the studies of Edward C. Pope and peers. There are characteristics of the fish that were widely accepted for many years before being disproven only in the last decade. For example, the ocean sunfish was once considered planktonic in habit, and to have very limited swimming ability (MacConnaughey, 1974, p. 170). Now, researchers are aware of the powerful swimming ability of the Mola mola that allows them to travel relatively long distances (Pope et al., 2010, p. 478). This paper will present the findings of these researchers including the animal’s characteristics, distribution, threats, and importance to the ecosystem. Taxonomy The ocean sunfish is a member of the order, Tetraodontiformes (Pope et al., 2010, p. 473). Other Tetraodontiformes include Tetradontidae, or pufferfishes and Diodontidae, or porcupine fish. The sunfish belongs to the family, Molidae and the genus, Mola. The family Molidae also contains the Masturus lanceolatus and the Ranzania laevis (Shapiro, 2013). The genus Mola contains the Mola mola and the closely related Mola ramsayi, the southern sunfish (Pope et al., 2010, p. 473). Body Structure Upon first look at the Mola mola, the animal appears to have its tail chopped off (Shapiro, 2013). The caudal fin is lacking on the Mola mola; a lobe called the clavus is present instead. The lack of a caudal fin led to the original perception that the fish were poor swimmers. The sunfish use other ways to swim through the water (Pope et al., 2010, p. 472). Powerful dorsal and anal fins are the fish’s primary means of locomotion. Mola mola are capable of horizontal movements, and deep-water dives (Pope et al., 2010, p. 474). The mola have massive dorsal and ventral fins and have small pectoral fins (Cardone, 1992, p. 128) (see Figure 1). Vision Many studies of the Mola mola’s eyes have concluded that their visual system is well-developed. This is deduced by researchers such as Pope from the fact that the Mola mola have massive eyes. Large eyes often indicate that vision is very important to the species. The size of the sunfish’s eyes leads to the conclusion that vision is a vital resource for the animal. Their eyes have also shown the ability to move around. This gives the sunfish a wider perspective that allows them to avoid predators and find prey (Pope et al., 2010, pg 477). Diet The Mola mola feeds mostly on gelatinous zooplankton, such as jellyfish. The ocean sunfish has unique beak-like teeth that they use to break apart the stingers of the jellyfish (Pope et al., 2010, p. 475). The purple-striped jellyfish attracts many Mola mola to the Monterey Bay in California (Cardone, 1992, pg 128). There is a question, however, how much the sunfish truly relies on the gelatinous zooplankton, because of their deep-diving behavior, which makes determining the total diet difficult (Pope et al., 2010, p. 475). The Mola mola have also been documented eating leptocephalus eel larvae. There is also a possibility that the sunfish eat some squid, as they have been captured by fishermen using squid as bait (Pope et al., 2010, p. 476). Reproduction Mola mola are the most fecund of all vertebrates (Pope et al., 2010, p. 480). Female sunfish can hold several hundred million eggs at one time (Shapiro, 2013). There is very little evidence available on spawning areas or spawning seasons. The spawning season in the Western Pacific is thought to be from August to October. Lifespan and the reproductive age of the Mola mola is unknown. The eggs must be very small because of the large number of eggs female sunfish hold. This is remarkable because the larvae grow from about .25cm to the 11 foot creatures they become, a lifetime increase of 60 million times (Pope et al., 2010, p. 480). Distribution Mola mola are found in temperate and tropical regions. The species has been found in the Mediterranean, North Atlantic, South Atlantic, Gulf of Mexico, and the West Pacific (Pope et al., 2010, p. 474). There are studies that show the migration of sunfish to temperate regions in the summer months. A 2008 study placed tags on multiple Mola mola, which showed an increased latitude in the winter months and a decreased latitude in the summer months. The sunfish traveled an average of 19.8km per day (Pope et al., 2010, p. 478). A study in the Northeast Atlantic showed estimates of 12,702 sunfish in the summer and 8,223 sunfish in the winter. This was the first study of sunfish in this region (Breen, 2017). The fish are seen with some regularity in California kelp forests but rarely found in the Florida Keys and Caribbean (Cardone, 1992, p. 128). South African waters have recently been determined the only known year-round habitat suitable for Mola mola (Pope et al., 2010, p. 479). Predators and Parasites Sea lions, orcas, and large sharks have been documented preying on the fish. Sea lions rip off the fins and beat their bodies against the surface to tear through the mola’s tough skin (Pope et al., 2010, p. 481). The decline of shark populations due to industrial fishing may lead to population growth of the Mola mola. Sunfish attract many smaller fish that form symbiotic relationships, that are often parasitic. There can be as many as 54 different species living off a single Mola mola at one time (Pope et al., 2010, p. 482). Organisms live on the outside and inside of fish; small shrimp have been photographed living inside the mouths of the sunfish (Cardone, 1992, p. 129). One of the most documented behaviors of the Mola mola is the basking on the surface by the species. The reason that the sunfish basks is heavily debated. One common explanation of the basking behavior is warming from the sun after diving into deep water. The other common explanation of basking is so that they can be cleaned of parasites by seabirds and fish near the surface (Pope et al., 2010, p. 481). Human Threats Most countries have no market for catching the ocean sunfish, however, Japan and Taiwan have been known to catch the fish for decoration purposes. There are high levels of incidental bycatch in the Mediterranean, Californian, and South African waters (Pope et al., 2010, p. 472). At a South African tuna fishery, Mola mola was caught at a rate of .29 sunfish for every 1000 hooks set. At the Cape horse mackerel water trawl fishery in South Africa, Mola mola accounts for 51% of all bycatch. One California swordfish fishery catches more Mola mola than they do swordfish (Pope et al., 2010, p. 482). Curving Threats The conservation status of the Mola mola is vulnerable and some countries have taken steps to reduce the bycatch rates of the sunfish. Spanish gillnets were a huge threat to Mola mola. The Spanish government closed fisheries that used gillnets in 1994. The Moroccan government passed a law in 2007 that stopped the use of gillnet fishing. The Moroccan fleet that forced the law to be set in place incidentally caught an average of 36,450 Mola mola per year (Pope et al., 2010, p. 482). Conclusion Relatively little is known about the ocean sunfish, but with each passing day, our understanding of the organism grows. As research technology advances, such as the use of tags and satellite tracking, Californian researchers such as Tierney M. Thys and company hope to expand their knowledge of the Mola’s migration patterns and site fidelity (Tierney et al. 2017). The mysterious, alien-like fish shows remarkable adaptations including basking and swimming using uniquely powerful dorsal and anal fins (Pope et al., 2010, p. 474). The incidental bycatch rates of the sunfish are alarming and more countries should follow the lead of Spain and Morocco and make gillnet fishing illegal in order to protect the fish (Pope et al., 2010, p. 482). References
Breen, P., Cañadas, A., Cadhla, O. Ó, Mackey, M., Scheidat, M., Geelhoed, S. C., . . . Jessopp, M. (2017). New insights into ocean sunfish (Mola mola) abundance and seasonal distribution in the northeast Atlantic. Scientific Reports, 7(1). doi:10.1038/s41598-017-02103-6 Cardone, B. J. (1995). The Fireside diver: an anthology of underwater adventure. New York: Aqua Quest Publications. MacConnaughey, B. H. (1974). Introduction to marine biology. Pope, E. C., Hays, G. C., Thys, T. M., Doyle, T. K., Sims, D. W., Queiroz, N., . . . Houghton, J. D. (2010). The biology and ecology of the ocean sunfish Mola mola: a review of current knowledge and future research perspectives. Reviews in Fish Biology and Fisheries, 20(4), 471-487. doi:10.1007/s11160-009-9155-9 Shapiro, L. (n.d.). Ocean Sunfish - Mola mola - Overview. Retrieved October 29, 2017, from http://eol.org/pages/213810/overview Thys, T. M., Hearn, A. R., Weng, K. C., Ryan, J. P., & Peñaherrera-Palma, C. (2017). Satellite Tracking and Site Fidelity of Short Ocean Sunfish, Mola ramsayi, in the Galapagos Islands. Journal of Marine Biology, 2017, 1-10. doi:10.1155/2017/7097965 By Justin H. Lor Abstract This paper, first, gives a brief description of the anatomy and important biological characteristics of the American Lobster, Homarus americanus. It then summarizes key characteristics exhibited in their most densely populated regions of the east coast. A lens is focused on the management practices of the American Lobster fishery. The success of one of the world’s most valuable fisheries followed by a recent decline in species abundance is looked at through multiple perspectives. Resource management, climate change and general predictable changes in ecosystem variation are all analyzed to explain population rise and fall. Future implications are analyzed. In conclusion, key elements of this research are reiterated to form a compact argument to why the American Lobster Fishery is in danger of experiencing a steep decline in population numbers and resulting loss of millions of dollars in revenue. Anatomy Lobsters have a hard-outer shell known as an exoskeleton. The outer parts of the body include a tail fin, antennae, walking legs, crusher claw, pincher claw and a long narrow body segment (see Graphic 1). The blood is often clear or slightly grey and circulated though large blood vessels by a heart located behind the lobster’s stomach. On the thorax section are gills that capture oxygen from the surrounding water. Lobsters have two compound eyes on the front region of their head. The antennae are used as feelers. Lobsters also have chemosensory antennules much like the nose of a human. The crusher claw or chelipod is used for crushing prey and as such is the larger of the two claws with a rounded surface. The smaller claw is more like a knife used for manipulating the previous crushed prey into the mouth. This pincher claw is like a spike. Pereiopods line the underbelly. These features are used as walking legs and are separated into two distinct types. The pereiopods located closer to the claws have sensors almost like taste buds. The rear periods function mostly as walking legs. There are various other features and complex structures within the lobster but for this research are mostly irrelevant (Lobster Institute at The University of Maine, 2017). Habitat Homarus americanus resides along the eastern seaboard of the United State of America. Ranging from Newfoundland, Canada all the way to North Carolina, USA. The species is primarily found on the continental shelf waters shallower than 50 meters. They are sometimes found at depths up to 700 meters but this is rare. This area comprises one of the most lucrative fisheries in the US. In 2006, the fishery was estimated to be valued at $386 million. The Gulf of Maine is where most commercial catch occurs and these harvests are most successful close to shore. Spatial distribution is largely dependent on both the size and sex. Seasonal and climatic changes also affect distribution but in most cases the smaller individuals are found closer to shore where larger lobsters can be found in deeper waters. Population density distribution is also likely subject to a variety of environmental factors including the opportunity to find available shelter, specific pH and salinity range, bottom temperature, sediments, and spatial variables such as latitude and longitude (Chang et al., 2010). Fishery Management The current state of the American Lobster fishery in New England is in decline. Despite strict size requirements being implemented to ensure a healthy population and deter overfishing the population is still experiencing losses. Small sized lobsters are illegal to harvest. They are further protected by trap requirements that force lobster fisherman to install vents on all lobster traps to allow smaller lobsters the opportunity of escape. There are also other laws that further eco-regulate the impacts of lobster fishing. To reduce by-catch and harm to species other than the intended lobster further regulations are being implemented. These regulations have made being a lobster fisherman an increasingly difficult prospect but have allowed the fishery to remain vital (Morrissey et al. 2015). Records dating back to the 1800s indicate a decline in overall species number. In 2012, the fisherman harvesting from the Gulf of Maine experienced record catches. This abundance of lobster flooded the market and subsequently had an adverse effect on industry sales. With supply greater than demand the price of the lobster dropped. This drop (about $1-$2 per pound) meant large financial losses for industries selling thousands of pounds at a time. The decline in overall species number in conjunction with easier harvestability (due to technological advances and refined techniques) contributes to a steep decline in any fishery or resource management effort. New markets are continually being created and expanded to allow for fast distribution of the oversupply of lobster. Overseas vendors are taker greater interest further threatening the species (Morrissey et al., 2015). Canadian/U.S. lobstermen perception of current ocean health To enhance communication and knowledge sharing within the community of concerned lobster fisherman, a one-and-a-half-day town meeting was held in Saint John, New Brunswick on March 20 and 21. This meeting brought together people of various sectors and geographic regions connected to the American Lobster Fishery. Attendees included people from New Brunswick, Maine, Massachusetts, Prince Edward Island and Nova Scotia totaling about 65 people. Among some of their concerns were issues like changing water temperatures, ocean acidification and marine pollution. The Lobster Institute at The University of Maine (2015) reports, “The lobstermen in attendance suggested priorities should include: a cross-border effort to further advance studies on ocean acidification; training in quality control for all handlers beyond the boat, and several others” (p. 3). Among the attendees of this Lobstermen town meeting was Maine State Senator Chris Johnson who cited findings by the Ocean Acidification Commission. This commission, co-chaired by Senator Johnson, recommends monitoring acidity changes more aggressively and increased study on the impacts of ocean acidification on species like lobster. He also stated that Maine will call for a $3 million bond referendum to carry out these proposed management efforts. (Lobster Institute at The University of Maine, 2015). Potential threats Climate change and the resulting ocean acidification is an obvious threat to efforts to the American Lobster Fishery. With so many changes forecasted as a result of the warming of Earths Ocean and atmosphere the lobster is among the many threatened species. Shell forming species like lobster are threatened from ocean acidification but this is only one of the changes that will challenge their survival. As noted earlier in this report the Lobster ecosystem is comprised of various elements that need to remain in balance for their continued survival. Things like pH, temperature and salinity are sure to change as global ice caps melt and sea levels rise and warm. Future Implications Countless fisherman lives will be among the many people impacted by the downfall of the American Lobster Fishery. With many efforts by fisherman, senators and concerned scientists and citizens the potential to sustain the Homarus americanus is possible but great threats loom in the wake of an unstable global system. Millions of dollars in resource value is likely to degrade. The lobster has proven to be a hardy survivor and great resource for hundreds of years though so hopefully the changes will not be too drastic. Importance This report draws from multiple sources to connect various reports with a common interest of many millions of people. The American Lobster has been an iconic species and a trophy to many. It even appears in art and folklore for many cultures (The Lobster Conservancy, 2004). In my life and many others, it has served as a centerpiece at lavish dinners and family gatherings. This hardy crustacean has proven itself to be one of the most valuable resources in the US. Lobster, to many, is a delicacy and is sought after from far reaches. It will be a great economic loss if the management of this vital resource fails. There are many good things being done to sustain the population but climate change seems to cast a large shadow over these efforts. Summary In conclusion of this report I propose that the American Lobster Fishery is in great danger of experiencing a steep decline. There are many great efforts to sustain the population but the vital elements of the lobster habitat are all likely to change with warming water temperature and rising sea levels. References
Chang, J.H., Chen, Y., Holland, D. & Grabowski, J. (2010). Estimating spatial distribution of American lobster Homarus americanus using habitat variables. Marine Ecology Progress Series, 2010, volume 420, 145-156. Retrieved from http://www.int-res.com/articles/meps2010/420/m420p145.pdf Morrissey, J., Sumich, J., & Pinkard-Meiser, D. (2015). Introduction to the Biology of Marine Life, Eleventh Edition. Pages 161 & 162 The Lobster Conservancy. (2004). Sustaining a thriving fishery through science and community - Lobster Biology. Retrieved from: http://www.lobsters.org/tlcbio/biology.html The University of Maine, Lobster Institute. (2015). Canadian/U.S. Lobstermen’s Town Meeting – Full Transcript. Retreived from: http://umaine.edu/lobsterinstitute/files/2015/07/Final-Transcript-2015.pdf The University of Maine, Lobster Institute. (2017). Life of the American Lobster – Anatomy & Biology. Retrieved from: http://umaine.edu/lobsterinstitute/education/life-of-the-american-lobster/anatomy-biology/ By Aaron Tucker Litwiler The Atlantic coast salt marshes of the United States are incredibly biodiverse and are home to many unique species of flora and fauna. One such species that calls this ecosystem home is the diamondback terrapin, Malaclemys terrapin. This turtle belongs to the family Emydidae which includes many common North American turtles such as box turtles, spotted turtles, and sliders (Seidel and Ernst). However, unlike the rest of the American turtles in this family, diamondback terrapins reside in the brackish waters of North America’s eastern coastline. The diamondback terrapin can be found in salt marsh, tidal flats, and estuaries from Cape Cod, Massachusetts down to Corpus Christi, Texas (Roosenburg, Willem). These turtles are known to prefer areas of low marsh that are frequently flooded, however they can be found in deeper grass beds, bays, and other deeper bodies of water. The salt marsh habitat is characterized by saltmeadow cordgrass (Spartina alterniflora), black needle rush (Juncus romerianus), and sea oxeye (Borrichea frutescens) (ATHENS, GEORGIA). The diamondback terrapin shares this habitat with countless species of fauna including Atlantic blue crabs (Callinectes sapidus) and common marsh snail (Melampus bidentatus), both of which fall prey to the terrapin (ATHENS, GEORGIA). The diet of diamondback terrapin mostly consists of snails, crabs, and mussels; however, they have been known to feed on aquatic worms, fish, insects, and even decomposing flesh. Large, more matured terrapins typically consume larger prey such as fiddler crabs, Uca pugilator, and the aforementioned Atlantic blue crab. Young terrapins and smaller males prey on snails such as marsh periwinkles, Littoraria irrorate, and the common marsh snail (ATHENS, GEORGIA). The difference in prey size amongst mature turtles is due to sexual dimorphism. Female diamondback terrapins are considerably larger than males of the species and have broader heads and larger grinding surfaces to accommodate larger prey selection. Male terrapins reach a maximum carapace length of 5.5 inches while females can grow twice that size maxing out at 11 inches of carapace length. Female terrapins also have broader heads, shorter tails, and taller shells. The latter two characteristics are due to the reproductive requirements of female turtles (Seidel and Ernst). Despite their sexual dimorphism, all diamondback terrapins share traits that are characteristic of the species. Several variations can occur amongst the seven subspecies terrapins such as carapace shape, shape of keel knobs, head and neck markings, and shell pigmentation, however they all share the same general appearance that sets diamondback terrapins apart from other species (Seidel and Ernst). They are excellent swimmers with large, stocky hind legs and smaller forelegs. Though terrapins inhabit saline environments they do not possess flippers like their sea turtle cousins; they have webbed feet instead. These turtles are flat-bodied with a grey, keeled carapace that has light and concentric markings. The plastron of this species lacks a hinge, is yellow in color, and is connected to the carapace by a flat bridge (Seidel and Ernst). Terrapins have grey or black skin that can be accompanied by dark speckling or mottling. Terrapins have adapted to the saline environment due to their development of lachrymal salt glands that help the turtles to expel salt (Gosse). Male terrapins have longer tails in order to grasp the female during mating as well as to accommodate presence of larger reproductive organs within their cloaca. Diamondback terrapins are oviparous, meaning the females must carry and lay their eggs before hatching. Consequently, females are larger in both carapace length and depth in order to accommodate a full clutch of eggs. Females lay their eggs in open, dry, sandy soil where they deposit anywhere between four to twenty-three eggs. Females can produce up to three clutches of eggs between early spring and late summer, however, they return to the same beach every year to lay (National Aquarium). The sex of the clutch is determined by the temperature at which they are incubated, much like sea turtles. The survivorship of the hatchling turtles is affected by factors such as the microclimate of the nesting area as well as the turtles’ energy store and nest predation (Seidel and Ernst). As a consequence of these, nest survivorship is typically between one and three percent. Raccoons are the primary consumer of turtle eggs, and are a large threat to their population. Raccoons are very tolerant of human development, and even flourish in developed habitat. These human developments are a threat to all brackish and saltwater turtles. The development of shorelines decreases breeding grounds for turtles along with the erosion control that is put to use. Bulkheads block turtles from accessing the dry sand, and the grasses that are planted tangle hatchling turtles and penetrate eggs. Crab pots have also been known to kill turtles by attracting curious turtles and trapping them underwater until they drown (Roosenburg, Willem) . Considering these threats to the diamondback terrapin, it is not hard to understand why they are threatened throughout much of their range. However, there is much that can be done to prevent the extinction of these turtles. Conservation efforts such as nest site protection, education of coastal communities, habitat protection, and crab pot restriction can increase the odds of survival of this incredible species (Roosenburg, Willem). Citations
Gosse, Andrew M. “Diamondback Terrapin (Malaclemys terrapin).” Savannah River Ecology Laboratory, University of Georgia, srelherp.uga.edu/turtles/malter.htm. Seidel, Michael E., and Carl H. Ernst. "A systematic review of the turtle family Emydidae." Vertebrate Zoology 67.1 (2017): 1-122. ATHENS, GEORGIA. Home range, habitat selection, and diet of the diamondback terrapin (Malaclemys terrapin) in a North Carolina estuary. Diss. The University of Georgia, 1998. Roosenburg, Willem M. "The diamondback terrapin: population dynamics, habitat requirements, and opportunities for conservation." New Perspectives in the Chesapeake System: A Research and Management Partnership. Proceedings of a Conference. CRC Publ. No. 137. 1991. “Diamondback Terrapin ( Malaclemys Terrapin).” National Aquarium, National Aquarium, aqua.org/explore/animals/diamondback-terrapin |
Biology of the Sea
|





 RSS Feed
RSS Feed
